|
See PAC (AOP ) for full details or visit Commission II.1 ĭownload the PDF version (letter size or A4) or A3 (PDF) or see earlier versionsĬheck out SPECIAL Chem Int Jan 2019 - International Year of the Periodic Table (IYPT) - with contributions by Jan Reedijk, Natalia Tarasova, G.J. For elements that lack isotopes with a characteristic isotopic abundance in natural terrestrial samples, the mass number of the nuclide with the longest confirmed half-life is listed between square brackets. In 1945 Glenn Seaborg identified lanthanides and actinides (atomic number >92), which are usually placed below the periodic table.The latest release of the Periodic Table (dated ) includes the most recent abridged standard atomic weight values released by the IUPAC Commission on Isotopic Abundances and Atomic Weights ( CIAAW), compiled as part of the 2021 Table of Standard Atomic Weights 2021. In that same year Englishman Cockroft and the Irishman Walton first split an atom by bombarding lithium in a particle accelerator, changing it to two helium nuclei. This was the complete basis for the periodic table. In 1932 James Chadwick first discovered neutrons, and isotopes were identified. English physicist Henry Moseley provided atomic numbers, based on the number of electrons in an atom, rather than based on atomic mass. He also transmutated a nitrogen atom into an oxygen atom for the first time. In 1914 Rutherford first identified protons in the atomic nucleus. Radiation is emitted during movement from one orbital to another. In 1913 Bohr discovered that electrons move around a nucleus in discrete energy called orbitals. In 1911 Rutherford and German physicist Hans Geiger discovered that electrons orbit the nucleus of an atom. In 1903 Rutherford announced that radioactivity is caused by the breakdown of atoms. In 1900 Bequerel discovered that electrons and beta particles as identified by the Curies are the same thing. John Townsend and Robert Millikan determined their exact charge and mass. Thomson first discovered electrons small negatively charged particles in an atom. In 1894 Sir William Ramsay and Lord Rayleigh discovered the noble gases, which were added to the periodic table as group 0. They discovered that beta particles were negatively charged. Marie and Pierre Curie started working on the radiation of uranium and thorium, and subsequently discovered radium and polonium. Thomson student from New Zealand Ernest Rutherford named three types of radiation alpha, beta and gamma rays. In 1886 French physicist Antoine Bequerel first discovered radioactivity. He predicted the discovery of other elements, and left spaces open in his periodic table for them. In 1869 Russian chemist Dimitri Mendeleev started the development of the periodic table, arranging chemical elements by atomic mass. In 1863 English chemist John Newlands divided the then discovered 56 elements into 11 groups, based on characteristics. In 1809 at least 47 elements were discovered, and scientists began to see patterns in the characteristics.  
In 1680 Robert Boyle also discovered phosphorus, and it became public. This was the first discovery of phosphorus. /PeriodicTableallcolor-58b5c82c3df78cdcd8bbb80f.png)
He heated residues from boiled urine, and a liquid dropped out and burst into flames. 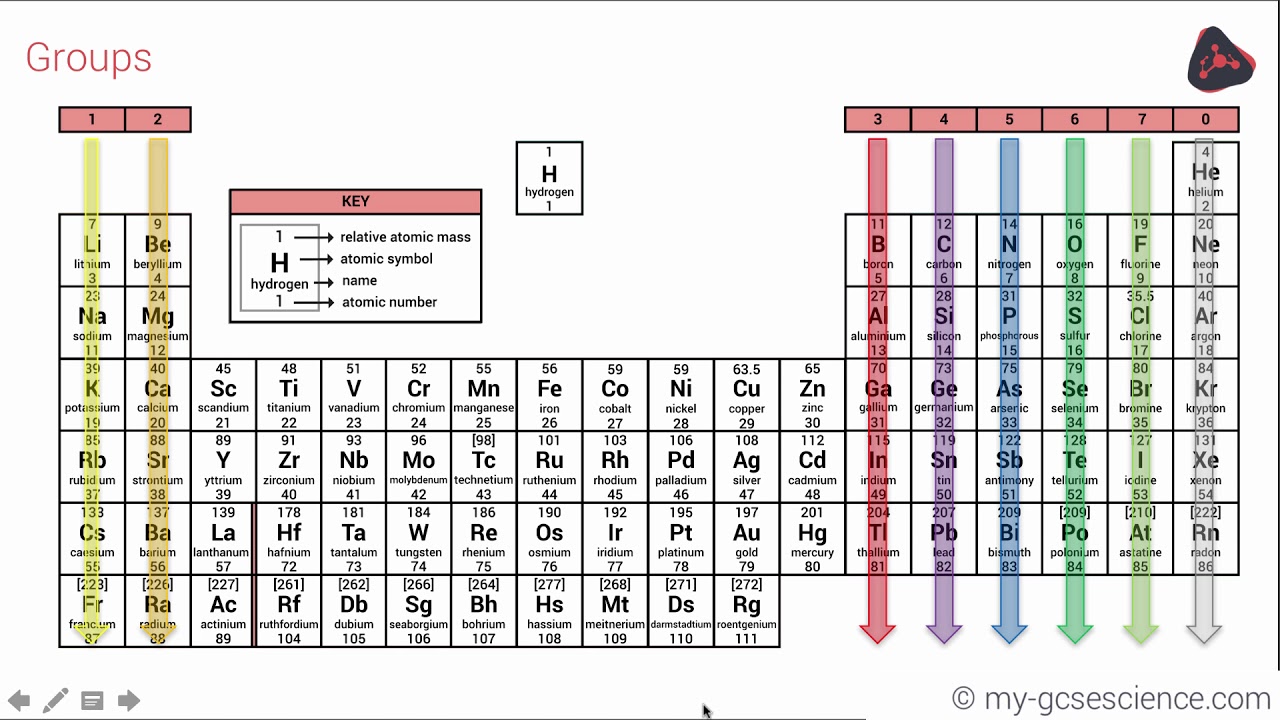
Periodic table history History of the periodic table of chemical elements In 1669 German merchant and amateur alchemist Hennig Brand attempted to created a Philosopher’s Stone an object that supposedly could turn metals into pure gold.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
